
The above are called immune checkpoint inhibitors (nivolumab, pembrolizumab, atezolizumab) that target and bind to PD-1 or PD-L1, denying PD-1 and PD-L1 linkage.
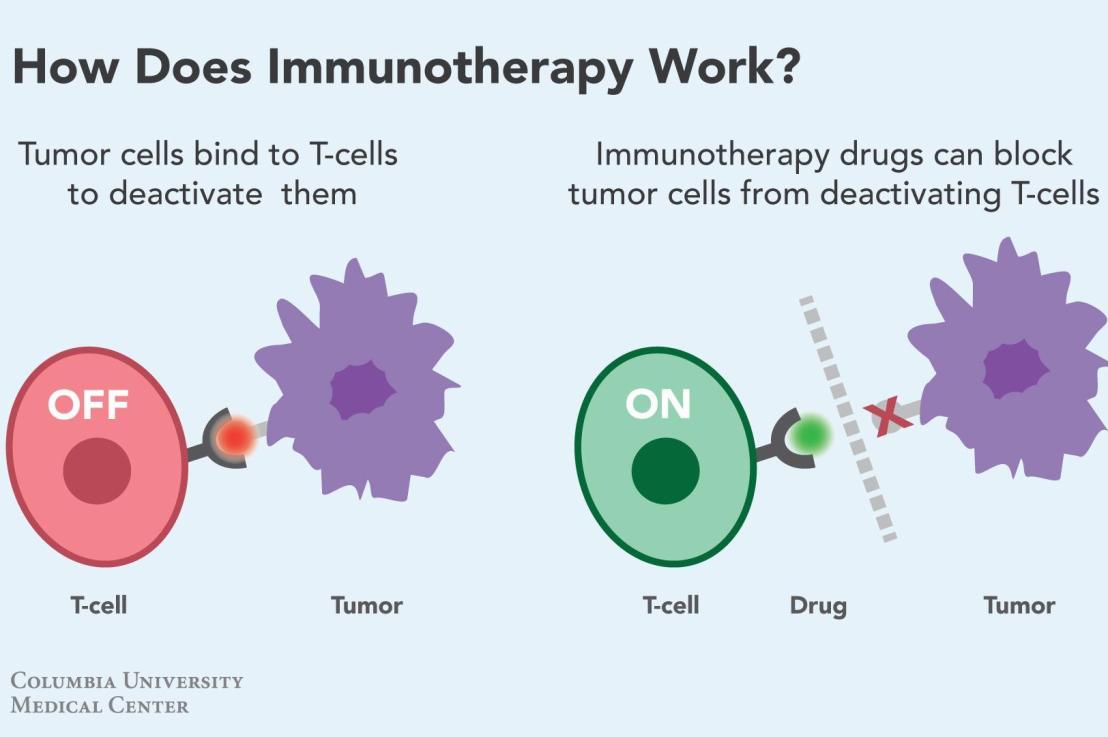
PD-1 is receptors on activated T-cells while PD-1 ligand, PD-L1, is present in normal cells (and also in some tumor cells).
Linkage of PD-1 and PD-1 ligand (PD-L1) will cause T-cells to be inactivated.
Nivolumab and pembrolizumab inhibit/ occupy PD-1 while atezolezumab inhibits/ occupies PD-L1.
Linkage of PD-L1 at tumor cells with PD-1 in immune T-cells will allow tumor cells to be ‘invisible’ and evade body immune system.
Tumour cells will be ‘visible’ and dealt by our own immune system (T-cells) once the PD-1 and PD-L1 binding sites are inhibited/ occupied by PD-1 or PD-L1 inhibitors, which disable the PD-1 and PD-L1 linkage.
It’s just as simple as the above cartoon.
Now, all patients will be happy to learn that it’s their own immune system that is fighting the cancer cells.
Immunotherapies and Results of Pivotal Trials
Immunotherapy treatment statistically improve median overall survival (OS) significantly in second line treatment of advanced/ metastatic lung cancer; reducing risk of death by about 30-40%.


They also have better overall response rate (ORR: partial response + complete response) and lower G3-4 toxicities when compared with chemotherapy (docetaxel).

Why Delay?
Many patient advocates (NGOs, pharmaceuticals and even healthcare providers) lament the government snail-tortoise pace in approving new immunotherapy (and other proven) treatments.
I heard our national pharmaceutical regulatory agency (NPRA) even requests ‘additional’ testing or data i.e one year product content stability (correct me if I’m wrong), which is not a common requirement for overseas regulatory agencies. (any new breakthrough medications need to waste additional one year before can be considered for approval?!)
Furthermore, most of those immunotherapy treatments already obtained approval from reputable Food and Drug Administration (FDA-USA) and European Medicines Agency (EMA-Europe).
More competitions
Faster approval of various immunotherapies will lead to more competitions amongst pharmaceutical companies and in the process will drive down the cost of immunotherapy treatment.
For example, atezolizumab was approved by FDA in 2016 but only obtained approval in Malaysia recently in second half of 2018.
As soon as it was approved in Malaysia, pembrolizumab (direct competitor) price fall from 20k+ per cycle (initially was even higher when it was launched) to sub 10k per cycle to match atezolizumab price.
So, can you see that your unnecessary delay is causing unnecessary high cost of treatment to patients who need it.
As you can see above, patients on average live around a year (in second line setting) and you already delayed approval of the treatment for 1-2 years. What gives?!
Cancer patients do not have so much time.
#drbhnglatest
One thought on “Advanced/ Stage 4 Lung Cancer: Approved Immunotherapy in 2nd Line Treatment (Part 1 of 2)”